Copyright © 1996, 1997, 2001
by Galen Daryl Knight and VitaleTherapeutics, Inc.
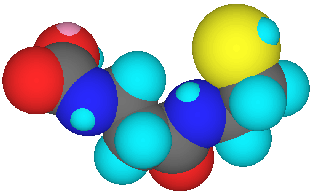
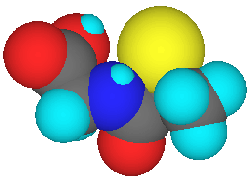
Tiopronin, A Truncated and Rearranged VitaleTheine?
Relative to vitaletheine (below left), tiopronin (below right) lacks the
methylene and nitrogen found adjacent to the carbamic acid moiety, its
amide is inverted, and the ethylene moiety between the sulfur and the amide
is rearranged to produce a methyl group alpha to the sulfur.
These and other structural changes to the vitaletheine modulators,
such as in levamisole, seem to cause dramatic
losses in potency at the same time that they generate analogues having
undesirable and even life-threatening side reactions, such as agranulocytosis,
hypersensitivity
reactions, and even death of normal tissue at the site of injection.
Still, the anti-rheumatic activities of tiopronin and levamisole are good
indicators that the vitaletheine modulators have potential in anti-autoimmune
diseases.
GO TO:

