Copyright © 2000, 2001 by Galen
Daryl Knight and VitaleTherapeutics, Inc.
PRODRUG FOR VITALETHEINE'S SULFENIC ACID
The vitaletheine modulator can be very unstable under high vacuum and in
the presence of matrices that contain metal ions capable of partially reducing
and aggregating the individual modulators. Thus, choice of matrix makes
an enormous difference in the mass ions obtained. A calcium salt of the
sulfenate-linked benzyl derivative produced the characteristic
mass ion when an inert target platform was used, but when stainless
steel is used an entirely different set of mass ions is observed:
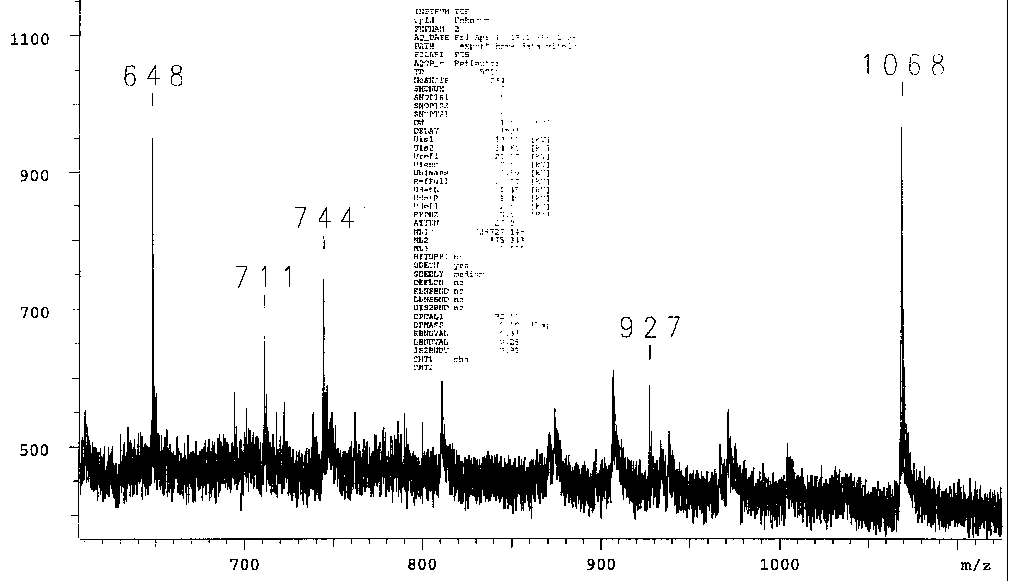
Similarly, when the synthetic process is contaminated even momentarily
with chlorine, the iodine monochloride being a stronger oxidant than iodine,
oxidation products can be formed with molecular weights about 16 mass units
higher than those for the authentic compound before oxidation. Sensitivity,
in a mass spectra illustrating these higher oxidation products, is further
enhanced by substances that tend to trap the targeted material and provide
a protective buffer between the target and the stainless steel support:
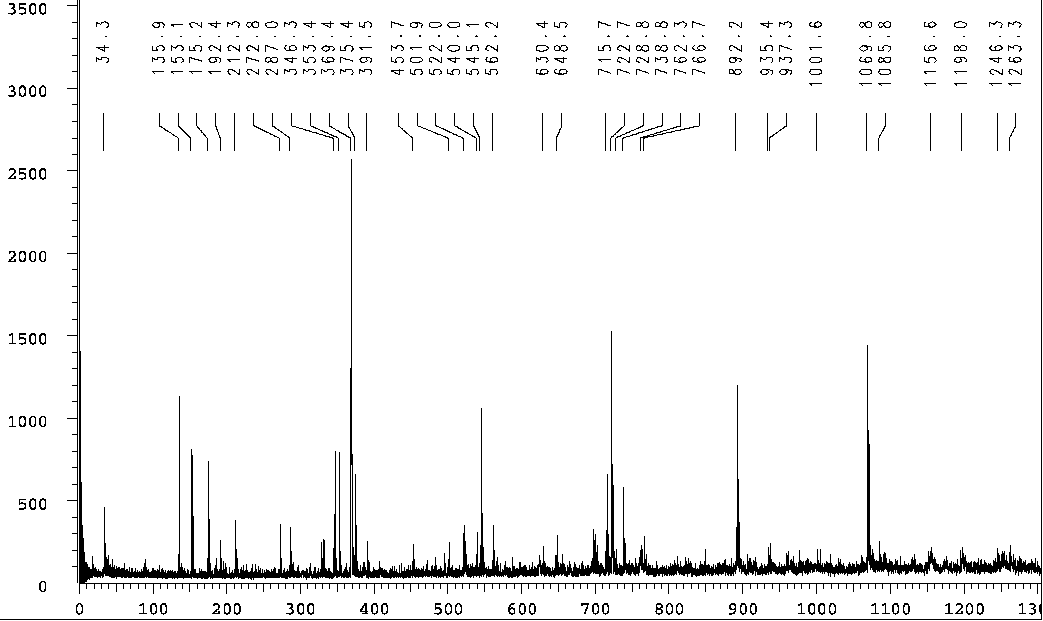
When this material is further modified ostensibly by reduction, the
higher mass ions (1068), associated with the authentic sulfenate-linked
benzyl carbamate dimer when desorbed from a stainless steel target, completely
disappear. Such gentle reduction with iodide in the mass spectrometer probably
does not reduce the higher oxidation states such as sulfinates [R-S(=O)-O-R2]
or sulfonates [R-S(=O)2-O-R2] or their respective
sufinic and sulfonic acids:
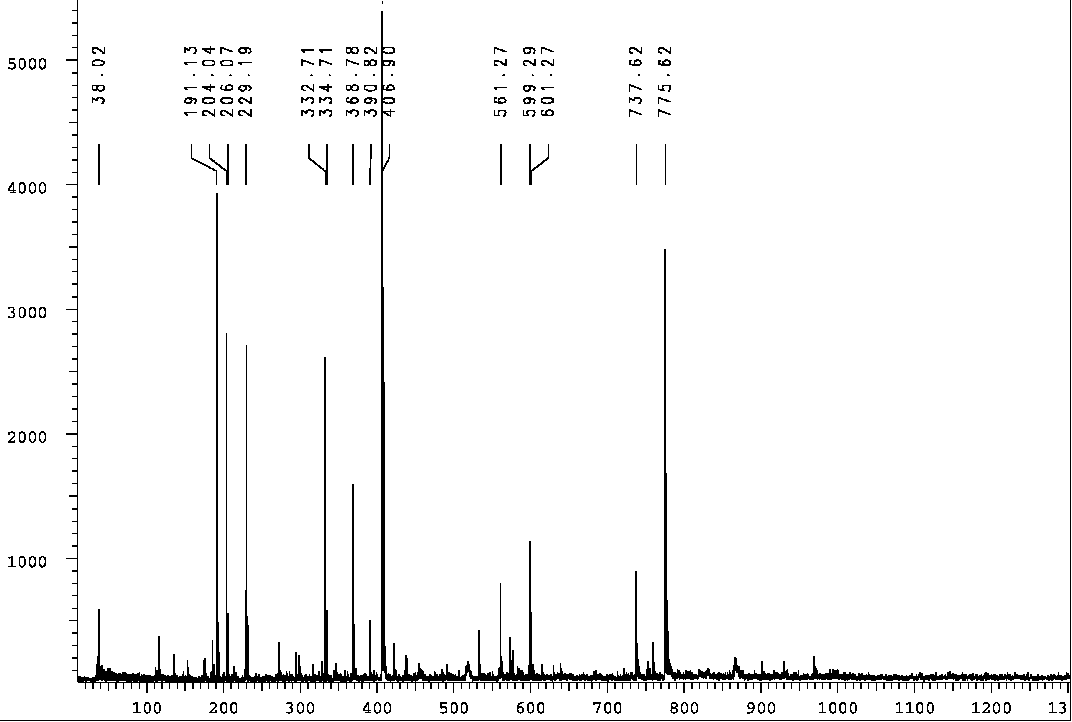
However, it is quite likely that iodide can reduce any sulfenic acid
groups in the matrix. Unfortunately, when iodide ions are made "acidic"
as in these MALDI mass spectrometer analyses, iodide can readily oxidize
to iodine. Under certain circumstances, such as extremes of pH, iodine
then can easily oxidize sulfenic acid groups to higher oxidation states,
a process conceivably catalyzed by metal ions that bind sulfur tightly.
Thus, the very high molecular weight species desorbed from the stainless
steel support with an "acidic" matrix readily disappear upon reduction
(of the metal?) with iodide ions, but interpretation of these results is
complicated by the oxidation of iodine under acidic conditions. Production
of higher oxidation states for sulfur (than those observed for the authentic
compound desorbing from the inert target support) are still possible under
these mildly reductive conditions through an oxidation by iodine catalyzed
by residual surface metal ions.
GO TO:

